Intermolecular Forces
Intermolecular Forces
In this lesson, students will first observe the changing charge cloud that surrounds a single polar molecule (HCl) and a single non-polar molecule (Cl2).
- Students will be able to distinguish intermolecular and intramolecular forces.
- Students will understand the differences between dipole-dipole interactions and induced dipole-induced dipole (London dispersion) interactions.
- dipole-dipole interactions
- dipole moment
- induced dipole-induced dipole interactions
- intermolecular forces
- intramolecular forces
- London dispersion forces
- van der Waals forces
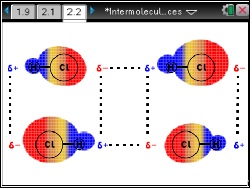
- In this lesson students will first observe the changing charge cloud that surrounds a single polar molecule (HCl) and a single non-polar molecule (Cl2).
- Next students are shown the lines of force that develop between adjacent HCl molecules and adjacent Cl2 molecules. These simulations will give students new insights into why molecules attract each other. They will also explore the different types of intermolecular interactions.
This activity was created using Lua. Learn more about Lua in TI-Nspire here.
Vernier EasyData,Vernier EasyLink and Vernier EasyTemp are registered trademarks of Vernier Science Education.

