NASA - Diving Down Deep
NASA - Diving Down Deep
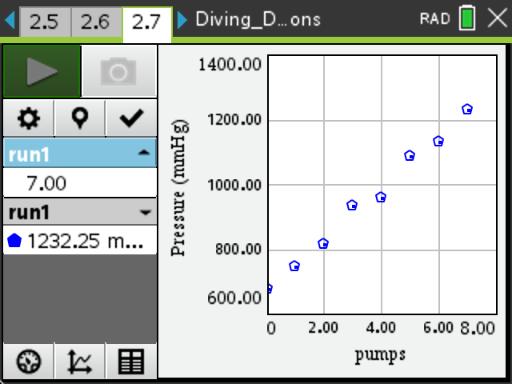
In this activity, students will explore a method for adding air to a gas cylinder diving tank, and determine the amount of gas needed by a support diver during a diving session.
Students will:
- explore and apply gas laws;
- add pressure to a bottle and determine the moles of gas added using the ideal gas law;
- create and interpret a mathematical model as it relates to the experiment performed; and
- related findings from the experiment to a real-world situation.
The NBL support divers assist the astronauts as they train for spacewalks in the NBL. The astronauts are in the water for up to six-hour sessions, and divers are in the water for up to two to three-hour sessions. The support divers use either a system of twin 6.95-liter tanks (containing a total of 2,832 liters of compressed nitrox) or a system of twin 11.1-liter tanks (containing a total of 4,530 liters of compressed nitrox).
Vernier EasyData,Vernier EasyLink and Vernier EasyTemp are registered trademarks of Vernier Science Education.

