Swimming with Acids and Bases
TI-Nspire™ CX CAS
Swimming with Acids and Bases
In this lesson, students will use a simulation to observe at the molecular level the difference between strong and weak acids and bases.
- Students will observe at the molecular level what makes a substance an acid or a base in water.
- Students will observe at the molecular level the difference between strong and weak acids and bases.
- acid
- base
- dissociation
- ion
- ionization
- strong acid/base
- weak acid/base
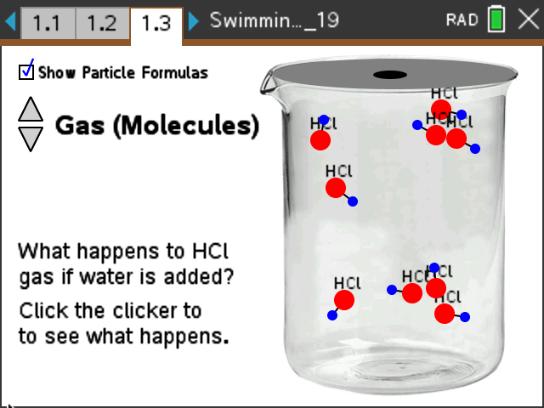
This lesson features simulations of a strong acid (HCl), a weak acid (HF), a strong base (NaOH), and a weak base (NH3) dissolving in water. Students will observe the changes that occur at the molecular level.
As a result, students will have a better understanding of:
- What makes a substance an acid or a base.
- What makes an acid or base strong or weak.
This activity was created using Lua. Learn more about Lua in TI-Nspire here.
TI-Nspire™ CX CAS
Vernier EasyData,Vernier EasyLink and Vernier EasyTemp are registered trademarks of Vernier Science Education.

