Vernier - Acid Rain
Science: Earth Science: Pollution
Science: Environmental Science: Pollution
Science: Biology: Ecology
9-12
60 Minutes
TI Connect™
TI Connect™ CE
TI Connect™ CE
TI-84 series
CBL™ 2
Sensor - pH
Sensor - pH
- Graph paper
- 250 ml and 100 ml beaker
- pH 7 buffer solution
- Dilute (0.02M) sulfuric acid
- Ring stand and clamp
- Straw
- Wash bottle with distilled water
- Lake water
- Ocean water or a synthetic mixture representative of ocean water
Vernier - Acid Rain
Activity Overview
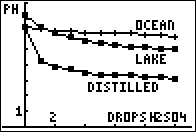
Students use the pH sensor to measure changes in the pH when CO2 dissolves in water. They also study the changes in the pH when sulphuric acid is added to water and identify water bodies most likely to be affected by acid rain.
Download Files
Lesson Files
Science: Earth Science: Pollution
Science: Environmental Science: Pollution
Science: Biology: Ecology
9-12
60 Minutes
TI Connect™
TI Connect™ CE
TI Connect™ CE
TI-84 series
CBL™ 2
Sensor - pH
Sensor - pH
- Graph paper
- 250 ml and 100 ml beaker
- pH 7 buffer solution
- Dilute (0.02M) sulfuric acid
- Ring stand and clamp
- Straw
- Wash bottle with distilled water
- Lake water
- Ocean water or a synthetic mixture representative of ocean water
iPad is a trademark of Apple Inc., registered in the U.S. and other countries.
Vernier EasyData,Vernier EasyLink and Vernier EasyTemp are registered trademarks of Vernier Science Education.
Vernier EasyData,Vernier EasyLink and Vernier EasyTemp are registered trademarks of Vernier Science Education.

